Lung Transplant
Cellvizio® is the real-time in vivo cellular imaging platform that enables physicians to monitor and assess the impact of COVID-19 in patients with lingering respiratory complications using proprietary advanced imaging technology.

COVID-19, the disease caused by the novel coronavirus, has created new and unprecedented obstacles for diagnosing and managing patients. As physicians and researchers learn more about COVID-19, the morphological aspect of this virus reveals that there are still many unknowns about its real time, in vivo characteristics and about the long-term impact it may have on the body.
There is an urgent need for a more accurate understanding of the virus’ morphological nature and effects on post recovery patients.
To address this important need, Mauna Kea Technologies is supporting research to determine if Cellvizio can be effective in severe acute and/or long COVID-19 patients by offering in-kind funding to researchers evaluating what role Cellvizio may play in the care management of COVID-19 patients. Interested clinical teams can submit their projects here.
Cellvizio can uncover vital information in four indications where the current diagnostic methods and tools leave ample room for research and development by leveraging probe- and needle-based Confocal Laser Endomicroscopy (pCLE/nCLE): pulmonary fibrosis, lung tissue microstructure, lung inflammation, and olfactory slits.
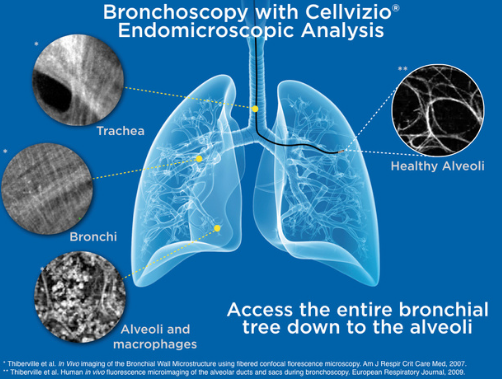
During a bronchoscopic procedure, Cellvizio’s advanced imaging technology may non-invasively, in vivo, and in real time assess areas of concern in COVID-19 compromised and recovering patients without incurring added risk while providing essential information to the physician.
In COVID-19’s active phase, Cellvizio can profile the neutrophil and macrophages within the alveolar space, which could be helpful in the early screening of critical illness, diagnosis, patient stratification, treatment monitoring and tailoring, or prognosis of COVID-19.
In the post-recovery phase, Cellvizio can assess lung damage such as fibrosis, microvascularization changes, and alveolar and capillary morphology damages at a microscopic level and non-invasively, which is significant for short and long-term patient management.
Pulmonary Fibrosis
COVID-19 patients are at high risk of developing pulmonary fibrosis and long-term impaired lung function at the peak of infection and during and after recovery.
Additionally, there is uncertainty surrounding the presence of other pulmonary infections and diseases in COVID-19 patients, such as ARDS, due to the sequential damage of decreased gas exchange in alveoli structure and function.
There is an urgent need for therapeutics to address the long-term effects of COVID-19, but there are definite risks and challenges in biopsying COVID-19 compromised patients.
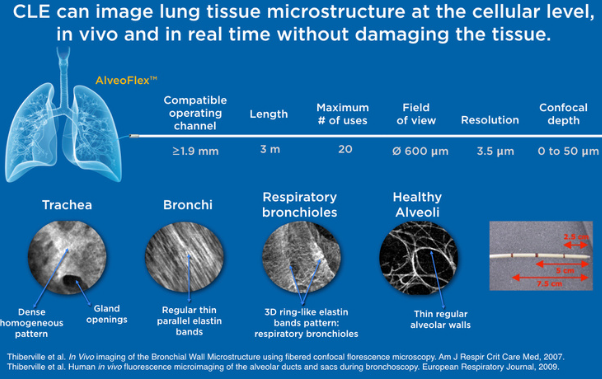
Currently, there is no in vivo and non destructive tool capable of assessing the grade of fibrosis with high specificity and accuracy. CLE is a high-resolution imaging technique that provides immediate feedback during bronchoscopy about the elastin fiber network of peripheral lung areas. Both the visceral pleura and fibrotic lung areas consist of elastin1 fibers and are, therefore, visualized with CLE.
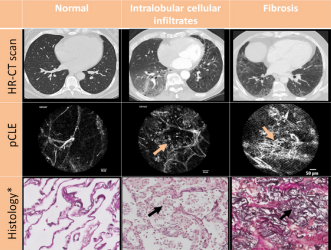
Thanks to its capability to image at a microscopic level, Cellvizio provides more specific information on tissue microstructure than CT2, leading to a clearer differentiation between fibrosis from infection.
In real time images enable physicians to make immediate patient management decisions based on the high specificity and clarity that Cellvizio provides.
Lung Tissue Microstructure
Despite widespread interest in the pathophysiology of the disease, relatively little is known about the associated morphologic and molecular changes in the peripheral lung of patients who die from COVID-19.
While ex-vivo, postmortem tissue analysis has shown differentiation between COVID-19 and the influenza virus infection, no diagnostic method currently exists to access microvascular structure in vivo and in real time.
There is pressing urgency to understand the pathogenesis of the severe acute respiratory syndrome coronavirus clade 2 (SARS-CoV-2), which causes the disease COVID-19. SARS-CoV-2 entry genes, which promote cellular entry, are most highly expressed in nasal goblet and ciliated cells within human airways.
While it is known that goblet cell metaplasia accompanies common pulmonary disorders that are prone to recurrent viral infections, the mechanisms regulating both goblet cell metaplasia and susceptibility to viral infection associated with lung diseases are not fully understood.
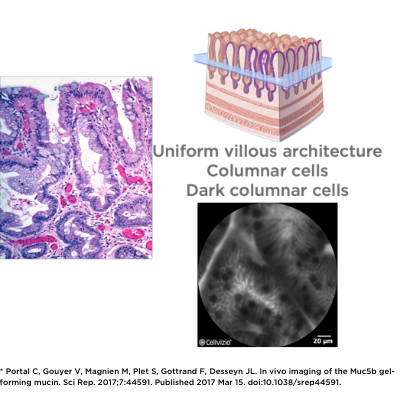
Cellvizio’s advanced imaging technology has the unique ability to visualize goblet cells and assess their makeup non-invasively and in vivo, as used in intestinal metaplasia.
Goblet cell imaging with CLE could provide physicians the necessary information to understand the pathogenesis of SARS-CoV-2.
Cellvizio’s ability to image tissue microstructure may offer a solution in the current obstacle of patient management, where much is still unknown about the morphologic and molecular changes in the peripheral lungs of COVID-19 infected patients.
Lung Inflammation
In several studies, post-mortem histology revealed lymphocytic endotheliitis in lung, heart, kidney, and liver, as well as liver cell necrosis, in patients suffering from COVID-193.
It is currently unknown whether vascular derangements in COVID-19 are due to endothelial cell involvement by the virus. No technique integrated in today’s standard of care is capable of imaging perivascular endotheliitis, in vivo and at a microscopic level, to reveal the widespread internal damage of COVID-19.
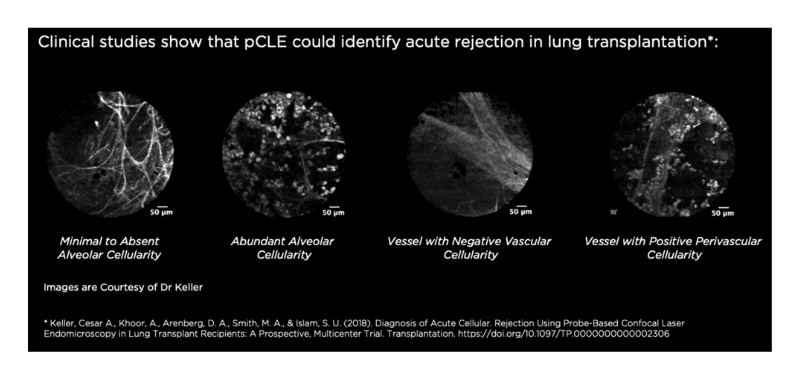
Probe-based Confocal Laser Endomicroscopy (pCLE) can assess acute lung rejection in lung transplantation by identifying perivascular inflammation.4 This existing ability of Cellvizio could be translated into observing the perivascular inflammation of COVID-19 compromised patients. Cellvizio pCLE could image perivascular endotheliitis, in vivo and at a microscopic level, to provide physicians with information about the internal spread of lymphocytic cells due to COVID-19.
Olfactory Slits
A large number of COVID-19-positive patients suffer from olfactory defects at early stages of the disease, where this symptom can remain post COVID-19 recovery5.
It has been shown that SARS-CoV-2 impairs olfactory slits6 and the olfactory slit area is difficult to reach in nasofibroscopy with the usual biopsy equipment. Moreover, biopsy is a destructive test. Today, a non-destructive, minimally invasive test does not exist to assess the olfactory slit in situ and in real time.
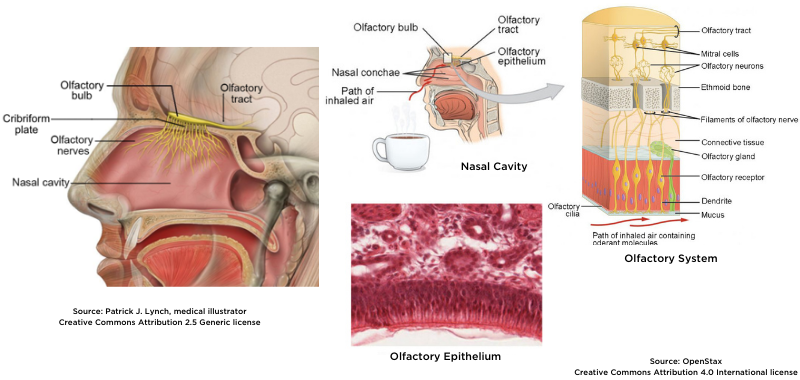
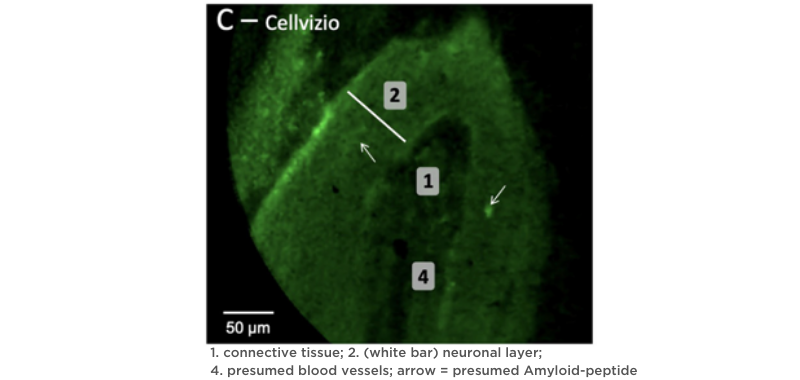
Because Cellvizio® is an in vivo and in real time endomicroscope, it can provide immediate answers while assessing olfactory cells7. Compared to the damage caused by biopsy, CLE causes no destruction of tissue.
Furthermore, Cellvizio® can be administered for this indication to an actively infected COVID-19 patient without incurring risk to the physician.
Cellvizio’s non-destructive nature provides an essential opportunity for the research of olfactory slits in COVID-19 patients so that the trajectory of this symptom can be eventually understood and managed.
References & disclaimers
1. Wijmans et al., Confocal Laser Endomicroscopy as a Guidance Tool for Transbronchial Lung Cryobiopsies in Interstitial Lung Disorder. Respiration 2019;97:259–263. DOI: 10.1159/000493271.
2. Kramer, T., Bronchoscopic needle based Confocal Laser Endomicroscopy (nCLE) as a Real-Time Detection Tool for Peripheral Lung Cancer. ERS 2020, Presented 2020 Sep.
3. Endothelial cell infection and endotheliitis in COVID-19, Published Online April 17, 2020 https://doi.org/10.1016/ S0140-6736(20)30937-5
4. Keller, Cesar A., Khoor, A., Arenberg, D. A., Smith, M. A., & Islam, S. U. (2018). Diagnosis of Acute Cellular. Rejection Using Probe-Based Confocal Laser Endomicroscopy in Lung Transplant Recipients: A Prospective, Multicenter Trial. Transplantation. https://doi.org/10.1097/TP.0000000000002306
5. Altin F, Cingi C, Uzun T, Bal C. Olfactory and gustatory abnormalities in COVID-19 cases [published online ahead of print, 2020 Jun 23]. Eur Arch Otorhinolaryngol. 2020;1-7. doi:10.1007/s00405-020-06155-9
6. Coolen T, Lolli V, Sadeghi N, et al. Early postmortem brain MRI findings in COVID-19 non-survivors [published online ahead of print, 2020 Jun 16]. Neurology. 2020;10.1212/WNL.0000000000010116. doi:10.1212/WNL.0000000000010116
7. Etievant A, Monnin J, Lihoreau T, et al. Comparison of Noninvasive Imagery Methods to Observe Healthy and Degenerated Olfactory Epithelium in Mice for the Early Diagnosis of Neurodegenerative Diseases. Front Neuroanat. 2020;14:34. Published 2020 Jul 14. doi:10.3389/fnana.2020.00034
News
YOU MIGHT ALSO LIKE TO READ

Mauna Kea Technologies Announces the Results of its Combined General Meeting of June 4, 2026
Mauna Kea Technologies Announces New Publication in the UEG Journal Identifying “Triple Healing”, a Cellvizio®— Assessed Therapeutic Target Associated with Sustained Remission in Ulcerative Colitis

Mauna Kea Technologies Expands Presence in the Middle East with UAE and Turkey Regulatory Approvals and First CellTolerance® Commercial Installation
Find out more by downloading the
Cellvizio Brochure
